|

2009, Vol. 4 No. 2, Article 41
Disposition of Ceftriaxone in Goats (Capra hircus)
S. Tiwari, Swati, S. K. Bhavsar*, U.
D. Patel and A. M. Thaker
Department of Pharmacology and Toxicology, College of Veterinary Science
and A. H.,
Anand Agricultural University, Anand-388001, Gujarat (INDIA)
*Corresponding Author;
e-mail address: [email protected]
ABSTRACT
Pharmacokinetics of ceftriaxone was determined in goats after single dose intravenous and intramuscular administration at the dose rate of 20 mg/kg body weight. The drug concentrations in plasma samples were measured through High Performance Liquid Chromatography. Following intravenous administration, the apparent volume of distribution, area under curve, elimination half-life and total body clearance were 0.58 ± 0.04 L/kg, 77.51 ± 7.49 mg.h/mL, 1.50 ± 0.05 h and 4.50 ± 0.43 mL/min/kg, respectively. Following intramuscular administration, the peak ceftriaxone plasma concentration of 21.51 ± 0.61 mg/mL was observed at 0.5h and the drug was detected up to 12h. The apparent volume of distribution, area under curve, elimination half-life
& total body clearance following intramuscular injection were 0.53 ± 0.05 L/kg, 66.78 ± 4.90 mg.h/mL, 2.03 ± 0.09 h and 3.04 ± 0.34 mL/min/kg, respectively.
The intramuscular bioavailability of ceftriaxone was 59.0 ± 4.0 %. Ceftriaxone has favorable pharmacokinetics with moderate bioavailability in goats indicates that the drug can be used to treat susceptible infections in goats at the dose of 20 mg/kg.
KEY
WORDS
Pharmacokinetics, bioavailability, ceftriaxone, goats, intravenous, intramuscular.
INTRODUCTION
Ceftriaxone is a third-generation semi-synthetic bactericidal cephalosporin, resistant to various types of bacterial
β-lactamases. It is effective against a wide variety of Gram-positive, Gram-negative and some anaerobic bacteria including
enterobacteriaceae, Haemophilus influenzae, Streptococcus pneumoniae and other non-enterococcal streptococci (Neu et al, 1981; Brogden and Ward, 1988). It has rapid absorption, wide distribution in tissues as well as body fluids, with good tolerance after parenteral administration in human beings (Christ, 1991). The drug thus seems to be extremely useful in a variety of infections including meningitis, septicemia, pyoderma, colibacillosis, surgical prophylaxis and urinary tract, respiratory tract, wound, soft tissues and joints infections (Richards et al, 1984). Pharmacokinetics of ceftriaxone have been investigated in calves, mares, dogs, goats, sheep and camel (Guerrini et al, 1985; Soback and Ziv, 1988; Johal and Srivastava, 1998, 1999; Gardner and Aucoin, 1994; Rebuelto et al, 2002; Dardi et al, 2004; Ismail, 2005; Goudah et al, 2006; Goudah, 2008). Diseases like coliform septicaemia, pneumonia, colibacillosis and meningitis are major causes of neonatal mortality in goats where ceftriaxone can be used for treatment. In spite of the great potential for clinical use of this drug in veterinary medicine, the data on its pharmacokinetics in goats are limited. Therefore, the present study was planned to determine the pharmacokinetics of ceftriaxone in goats following single intravenous and intramuscular administration at the dose of 20 mg/kg of body weight.
MATERIALS AND METHODS
Experimental animals and drug administration
The experiment was conducted on six healthy female Surti goats (20-24 months of age) weighing between 20.1-31.5 kg. The animals were kept under constant observation for two weeks before commencement of the experiment at instructional farm of the college. The animals were examined clinically to evaluate health status and to rule out the possibility of any diseases. Each animal was housed in a separate pen and provided standard ration. Water was provided ad libitum. All animals were randomly allocated to receive either intravenous or intramuscular injection of ceftriaxone sodium (Vetaceph, Unichem Pharmaceuticals Ltd., India) at the dose rate of 20 mg/kg. A washout period of 3 weeks was observed between treatments.
Collection of samples
Blood samples (3 mL each) were collected through the intravenous catheter fixed in contra-lateral jugular vein into heparinized glass test tubes before administration and at 2, 5, 10, 15, 30 min and 1, 2, 4, 6, 8, 12, 24, 36 and 48hr after intravenous administration while blood samples were collected before administration and at 5, 10, 15, 30 min and 1, 2, 4, 6, 8, 12, 24, 36 and 48hr after intramuscular administration of the drug. Plasma was separated by centrifugation at 3,000 revolutions per min for 10 min at room temperature and stored at –20°C and assayed with in 24 h.
Bioassay of ceftriaxone and pharmacokinetic analysis
Plasma ceftriaxone concentration was determined by the high performance liquid chromatography (HPLC) with minor modifications (Hakim et al, 1988). The HPLC system (Merck-Hitachi LaChrom) consists of isocratic pump (L-7110) with an online degasser (L-7612), interface (D-7000), UV detector (7400), autosampler (7200), sample cooler (L-7200), chromatography data station software (D-7000) and multi HSM-manager. Chromatographic separation was done using Lichrocart RP-18 column (250 mm X 4 mm) at room temperature.
Samples (250 ml) were deproteinized by addition of acetonitrile (500 ml), vortexed for one minute followed by centrifugation for 10 min at 5,000 revolutions per minute. A clear supernant fluid was decanted in a glass insert (automatic sampler vessels) from which 50 ml was injected into the HPLC system. The mobile phase consisted of a mixture of buffer and acetonitrile (62:38). The buffer was prepared by dissolving 1.78 g of di-sodium hydrogen phosphate dihydrate and 1.0 g of N-acetyl -N, N, N-trimethyl ammonium bromide in 950 mL of Milli Q water, pH (7.0) was adjusted with orthophosphoric acid. Mobile phase was filtered through 0.45 m Millipore filter. Mobile phase was pumped through column at a flow rate of 1.0 mL/min, at an ambient temperature of 25°C. The eluate was monitored at a wavelength of 254 nm. All chemicals used in the present study were of HPLC grade.
Ceftriaxone standards (0.19, 0.26, 0.52, 1.68, 4.93, 14.94, 49.79, 76.59, 90.11, 100.12 mg/mL) were prepared by serial dilutions of stock solution of the pure ceftriaxone in drug-free plasma of goats. Calibration curve was prepared for drug concentrations ranging from 0.19 to 100.12 mg/mL and was used to quantify the drug concentration in samples. The calibration curve was prepared daily and it had a R2 value ≥ 0.99. The assay was linear for drug concentrations of 0.19 to 100.12 mg/mL. The lower limit of quantification of assay was 0.19 mg/mL. Different pharmacokinetic parameters were calculated using following equations as described by Gibaldi and Perrier (1982) and Notari (1987).
a) Half-life: distribution, elimination and absorption phases:
(i)
t1/2α
= 0.693/α
(ii)
t1/2β
= 0.693/β (iii)
t1/2 k(a)
= 0.693/
k(a)
b) AUC (0 -
∞), the total area under the serum drug concentration – time curve and AUMC, the area under the first moment of the serum drug concentration – time curve were calculated by trapezoidal rule.
c) Vd(area), the apparent volume of distribution:
Vd(area) = Dose (mg/kg) /
β X (AUC)
d) Vd(ss), the volume of distribution of drug at steady state:
Vd(ss) = Dose x AUMC / (AUC)2
e) ClB, the total body clearance of drug:
ClB =
β x Vd(ss) x 1000
f) MRT, the mean residence time:
MRT = AUMC / AUC
g) F, the fraction of drug absorbed after non-vascular administration:
F = t½β (I.V.) x AUC (I.M.) / t½β (I.M.) x AUC (I.V.)
RESULTS
Following intravenous and intramuscular administration, the data were best fitted to two-compartment and one compartment open model, respectively. The drug was detected in plasma up to 8 and 12 h following intravenous and intramuscular administration, respectively. Comparative disposition of ceftriaxone following single dose intravenous and intramuscular administration in goats is shown on semilogarithmic scale in Fig. 1.
Following intravenous administration the drug was rapidly distributed (t1/2α
: 0.12 ± 0.01 h; Vd(area): 0.58 ± 0.038 L/kg) and eliminated (t1/2β: 1.50 ± 0.05 h) from the body with a clearance rate of 4.50 ± 0.43 mL/min/kg. Following intramuscular administration, the peak plasma drug concentration was 21.51 ± 0.61 mg/mL (Cmax) at 0.5 h (Tmax) and the drug was detected up to 12 h. The drug was rapidly absorbed (t1/2 k(a) : 0.08 ± 0.01), widely distributed (Vd(area): 0.53 ± 0.05 L/kg) and slowly eliminated (t1/2β: 2.03 ± 0.09 h; ClB: 3.04 ± 0.34 mL/min/kg) following intramuscular injection. The bioavailability of ceftriaxone was 59.0 ± 4.0 % following intramuscular injection. Various pharmacokinetic parameters calculated from plasma concentration of ceftriaxone after its single dose intravenous and intramuscular administrations are summarized in Table 1.
DISCUSSIONS
Following intravenous administration of the drug, the drug concentration in plasma (0.32 ± 0.03 mg/mL) was measured at 8h. Johal and Srivastava (1999) reported similar observation in crossed bred calves, however Soback and Ziv (1988) reported lower trough concentration of 0.1 mg/mL at 10h post intravenous injection in neonatal calves. Peak plasma concentration (Cmax) of 21.51 ± 0.61 mg/mL was observed at 0.5 h following intramuscular injection, which is in agreement with observations reported in crossed bred calves (20.3 ± 0.92 mg/mL), goats (23.6 ± 1.2 mg/mL) and sheep (23.16 ± 2.94 mg/mL) (Johal and Srivastava, 1998; Ismail, 2005; Goudah et al, 2006).
Following intravenous administration, faster distribution (α: 5.77 ± 2.00 h-1) and slow elimination (β: 0.46 ± 0.02 h-1) indicated that the drug is rapidly distributed and then relatively slowly eliminated in goats. The elimination half-life of ceftriaxone was 1.50 ± 0.05 h. It is in agreement with the half life of ceftriaxone reported in calves (1.39 ± 0.04 h), sheep (1.7 h) and goats (1.44 ± 0.01 and 1.75 ± 0.02 h) (Guerrini et al, 1985; Soback and Ziv 1988; Ismail, 2005; Goudah et al, 2006). However longer elimination half-life of 2.57 ± 0.52 h was reported in camel (Goudah, 2008) may be due to slower body clearance (1.83 ± 0.16 mL/min/kg) of the drug in camel. The total body clearance of ceftriaxone following intravenous administration in goats was 4.50 ± 0.43 mL/min/kg which supports faster elimination of the drug in calves, sheep, goats and horses (Guerrini et al, 1985; Soback and Ziv 1988; Gardener and Aucoin, 1994; Ismail, 2005; Goudah et al, 2006). Elimination half life and total body clearance suggests faster elimination of cetriaxone in goats. The apparent volume of distribution of the drug following intravenous administration was 0.58 ± 0.04 L/kg, which is lower than the apparent volume of distribution of 1.19 ± 0.19 L/kg reported in cow (Johal and Srivatava, 1998).
Following intramuscular administration, the elimination half-life of ceftriaxone (2.03 ± 0.09 h) is similar to elimination half-life of ceftriaxone reported in cow calves (1.94 ± 0.12), sheep (1.77 ± 0.24) and goats (1.44) (Soback and Ziv 1988; Ismail, 2005; Goudah et al., 2006). The intramuscular bioavailability of ceftriaxone was 59.0 ± 4.0 per cent. However, higher bioavailability of ceftriaxone following intramuscular administration has been reported in cow calves (78%), buffalo calves (86.7%), goats (85%), sheep (83.6 ± 20.53%) and camel (93.42 ± 21.4%) (Soback and Ziv, 1988; Dardi et al, 2004; Ismail, 2005; Goudah et al, 2006; Goudah, 2008).
The success of antimicrobial therapy can be largely determined by integrating the pharmacokinetic and pharmacodynamics parameters like AUC/MIC and Cmax/MIC (Mckellar et al, 2004). The effective use of the antibacterial drugs against clinically important pathogens depends on designing dosages that attain a Cmax/MIC ratio > 8-10 and an AUC/MIC ratio > 100-125 (Lode et al, 1998). Thus values of AUC/MIC and Cmax/MIC after intramuscular administration were calculated using MIC90 (0.2 mg/mL) of ceftriaxone against
Salmonella, Escherichia coli and Pasteurella multocida isolates (Soback and Ziv, 1988). The calculated value of Cmax/MIC (107.55) and AUC/MIC (333.9) indicates that ceftriaxone may have excellent clinical and bacteriological efficacy against gram negative infections in goats. In conclusion, ceftriaxone can be used at the dose of 20 mg/kg to treat various susceptible infections in goat.
REFERENCES
-
Brogden RN, Ward, A. Ceftriaxone: A reappraisal of its antibacterial activity and pharmacokinetic properties, and an update on its therapeutic use with particular reference to once-daily administration. Drugs 1988; 35: 604-645.
-
Christ, W. Pharmacological properties of cephalosporins. Infection 1991; 19: 244-252.
-
Dardi MS, Sharma SK, Srivastava AK. Pharmacokinetics and dosage regimen of ceftriaxone in buffalo calves. Vet Res Commun 2004, 28: 331-338.
-
Gardner SY, Aucoin DP. Pharmacokinetics of ceftriaxone in mares. J Vet Pharmacol Ther 1994; 17, 155-156.
Gibaldi M, Perrier D. Pharmacokinetics. (2nd Edn.), Marcel-Dekker Inc., New York, 1982, pp. 45-109.
-
Goudah A, Shin HC, Shim AJ, Abd El-Aty AM. Charecterization of the relation ship between serum and milk residue disposition of ceftriaxone in lactating ewes. J Vet Pharmacol Ther 2006; 29, 307-312.
-
Goudah A. Pharmacokinetic parameters of ceftriaxone after single intravenous and intramuscular administration in camels (Camelus Dromedarius). Res Vet Sci 2008; 84, 483–489
-
Guerrini VH, Filippich LJ, Cao GR, English PB, Bourne DWA. Pharmacokinetics of cefaronide, ceftriaxone and cefoperazone in sheep. J Vet Pharmacol Ther 1985; 8, 120-127.
-
Hakim L, Bourne DW, Triggs EJ. High performance liquid chromatographic assay of cefotaxime, desacetylcefotaxime and ceftriaxone in rat plasma. J Chromatogr 1988; 424:111-117.
-
Ismail MM. Pharmacokinetics, urinary and mammary excretion of ceftriaxone in lactating goats. J Vet Med A : Physiol Pathol Clin Med 2005; 52, 354-358.
-
Johal B, Srivastava AK. Disposition kinetics and dosage regimen of ceftriaxone in crossbred calves. Acta Vet Hung 1999; 47, 243-248.
-
Johal B, Srivastava AK. Pharmacokinetics, urinary excretion and dosage regimen of ceftriaxone in crossbred cow calves following single intramuscular administration. Indian J Ani Sci 1998; 68: 1017-1019.
-
Lode, H., Borner, K., Koeppe, P.: Pharmacodynamics of fluoroquinolones. Clin. Infect. Dis. 1998; 27: 33-39.
-
Mckellar, Q.A., Sanchez Bruni, S.F., Jones, D.G. : Pharmacokinetic/pharmacodynamic relationships of antimicrobial drugs used in veterinary medicine. J. Vet. Pharmacol. Ther. 2004; 27: 503–514.
-
Neu HC, Merpol NJ, Fu KP. Antibacterial activity of ceftriaxone, a beta-lactamase-stable cephalosporin. Antimicrob Agents Chemother 1981; 19: 414-423.
-
Notari RE. Biopharmaceutics and clinical pharmacokinetics- An introduction. 4th Edn. Marcell dekker Inc. New York, 1987.
-
Rebuelto M, Albarellos G, Ambros L, Kreil V, Montoya L, Bonafine R, Otero P, Hallu R. Pharmacokinetics of ceftriaxone administered by the intravenous, intramuscular or subcutaneous routes to dogs. J Vet Pharmacol Ther 2002; 25, 73-76.
-
Richards DM, Heel RC, Brogden RN, Speight TM, Avery GS. Ceftriaxone. A review of its antibacterial activity, pharmacological properties and therapeutic use. Drugs. 1984; 27, 469-527.
-
Soback S, Ziv G. Pharmacokinetics and bioavailability of ceftriaxone administered intravenously and intramuscularly to calves. Am J Vet Res 1988; 49: 535-538.
Table 1: Pharmacokinetic parameters of ceftriaxone after single dose intravenous
and intramuscular administration (20 mg/kg) in goats
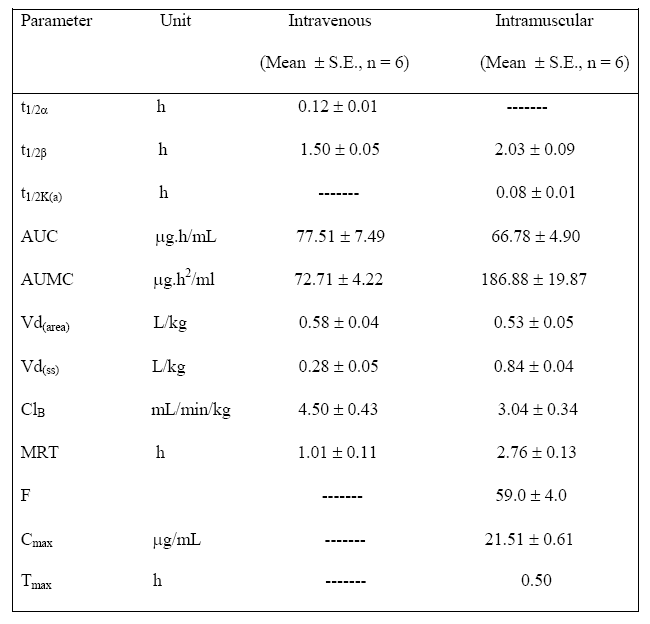
t1/2a:
half-life
of distribution phases; t1/2β:
elimination half life;
t1/2K(a):
absorption half-life;
AUC: total area under plasma drug concentration-time
curve; AUMC:
area under first moment of curve;
Vd(area): apparent volume
of distribution;
Vd(ss):
volume of distribution at steady state; Cl(B):
total plasma clearance; MRT: mean residence time;
F:
bioavailability;
Cmax:
maximum drug concentration; Tmax:
time of maximum observed concentration in plasma
Fig 1: Semilogarithmic plot of ceftriaxone concentration in plasma versus time following single dose intravenous and intramuscular administration at the dose rate of 20 mg/kg of body weight in goats. Each point represents mean ± S.E. of six animals.

|
 |




