|

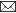
2010, Vol. 5 No. 1, Article 50
A Histo-Anatomical Study of Dorsal Lacrimal Gland
in Iranian River Buffalo
M. Shadkhast*1 and A. S. Bigham2
1Department of Basic Science,
2Department of Veterinary Surgery
Faculty of Veterinary Medicine, Shahre-kord University, Shahre-kord, Iran
*Corresponding Author;
e-mail address: [email protected]
ABSTRACT
Dorsal lacrimal glands (n=7) of Iranian river buffalos without apparent ocular disease were examined to compare the normal anatomical properties of these glands. All glands were characterized and measured (length and width). The gross anatomy of the dorsal lacrimal glands was similar, with the exception of a bipartite gland in cattle. A subset of the Iranian river buffalo samples (n=7) was sectioned for histological analysis. The histology of the dorsal lacrimal gland revealed tubulo-alveolar cells with basophilic vacuolated cytoplasm in Iranian river buffalo.
KEY WORDS
Lacrimal gland, River buffalo.
INTRODUCTION
Lacrimal glands are responsible for the
production of tear fluid that helps to maintain corneal health. These
glands contribute to the major proportion of tear in most of the
species. Examination of lacrimal glands of human beings, dogs, cats,
horses, pigs, rabbits, sheep, goats, guinea-pigs and single-humped camel
has been described in the literature (1-8). In general, the lacrimal
gland is tubuloalveolar structure that produces a mucoserous secretion.
Although similarities exist between the dorsal lacrimal and superior
gland of the third eyelid among species, differences exist in the
presence and composition between domesticated ungulates, cattle (7). The
histology of the lacrimal glands in cattle has been reported previously
(7, 9). However, description about the lacrimal glands of Iranian river buffalos is not available in the literature as per the author’s knowledge. The aim of this study was to describe the normal anatomical and histological details of Iranian river buffalos.
MATERIALS AND METHODS
The lacrimal glands of Iranian river buffalos (n=20; 12 male and 8 female) were obtained from local slaughterhouse. The glands were retrieved within 1 hour of slaughter.
Pathological examination of the eyes and adnexa revealed no apparent ocular disease. Length and width to the nearest millimeter were recorded for each gland and mean values were established for each group of glands.
The glands were fixed in 10% buffered formalin solution and processed to obtain 5 μm thick paraffin sections. Histological analyses were performed on randomly selected samples collected from seven Iranian river buffalos (five male and two female). Sections were stained with hematoxylin & eosin stain and examined by light microscopy for histological description.
RESULTS
Gross anatomy:
The average age of all the buffalos under study was 2.5±0.6 years. The dorsal lacrimal glands were present in Iranian river buffalos. The dorsal lacrimal glands were similar in appearance with
that of cattle with the exception of accessory lobe that was not
present in the glands obtained from Iranian river buffalos (Fig. 1).
The main body of the gland was flattened, oval, pale yellow and lobulated in gross appearance. These glands were located within
the fascia of the periorbit at the level of the dorsolateral aspect of the eye ball beneath the frontal bone. The inferior surface was slightly concave, fitting over the eye ball. No significant differences were seen in the length and width between both sexes.
Light microscopy:
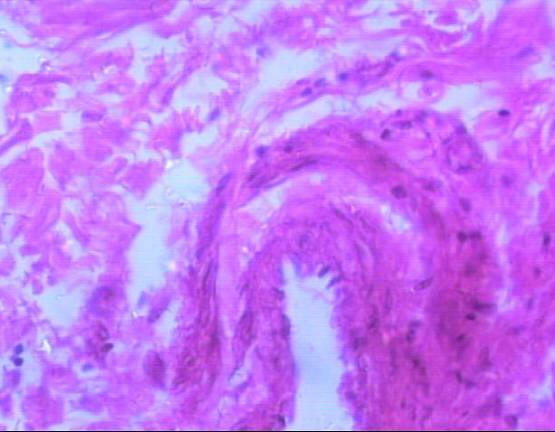
Histological examination of the lacrimal glands of Iranian river buffalo revealed similarities and differences between the species. The gland consisted of tubuloacinar units separated by dense sheets of connective tissue into lobules. Within a lobule, single sheets of connective tissue separated acinar and tubular units from each other. The acini were composed of tall pyramidal or columnar cells with small lumens; the tubules were bordered by short columnar cells with large lumens. The tubules were seen intermingled between the nests of acini. The buffalo acinar cells had basophilic, granular, vacuolated cytoplasm and appeared to be arranged in a loosely irregular fashion (Fig. 2, 3). Inter-lobular ducts with pseudostratified lining epithelium, veins and arterioles were found in connective tissue septae that separated lobes of the glands (Fig. 4).
DISCUSSIONS
The lacrimal glands of buffalo and cattle were basically similar in appearance. The gland had a lobulated appearance in both species of animals. It was interesting to find that the buffalo did not possess a bipartite dorsal lacrimal gland as seen in cattle (7). However, dorsal lacrimal glands of buffalo were longer as compared to cattle. The buffalo lacrimal glands were 81.5 mm in length (mean
81.5±0.41 SE) and 32.2 mm in width (mean 32.2±0.51 SE). Prince et al. (1960) reported the cattle’s dorsal lacrimal gland to be approximately 65mm in length and 36mm in width, however, age, breed and gender were not specified. There is an effect of ageing on the lacrimal system in humans (10) and rats (11, 12). To examine ageing effects in buffalo a longitudinal study was required which was beyond the scope of the present study. There was no significant gender difference anatomically or histologically in buffalo samples. In contrast to our observations, gender-related differences have been reported in rats, mice, guinea-pigs, rabbits and humans (13-15). Specifically, acinar lumens were reported to be larger in males than females which may be due to sex hormones (16).
REFERENCES
-
Allen M, Wright P, Reid L. The human lacrimal gland. A histochemical and organ culture study of the secretory cells. Arch Ophthalmol 1972;88(5):493-7.
-
Awkati A, al-Bagdadi F. Lacrimal gland of the camel (Camelus dromedarius). Am J Vet Res 1971;32(3):505-10.
-
Gargiulo AM, Dall'Aglio C, Coliolo P, Ceccarelli P, Pedini V. Complex Carbohydrate Histochemistry and Ultracytochemistry of the Sheep Lacrimal Gland. Anatomia, Histologia, Embryologia 2000;29(1):19-24.
-
Gillette TE, Allansmith MR, Greiner JV, Janusz M. Histologic and immunohistologic comparison of main and accessory lacrimal tissue. Am J Ophthalmol 1980;89(5):724-30.
-
Krochmalska L. Histological and histochemical picture of the lachrymal gland in guinea pig. Acta Theriologica;21(1-11):31-6.
-
Martin CL, Munnell J, Kaswan R. Normal ultrastructure and histochemical characteristics of canine lacrimal glands. Am J Vet Res 1988;49(9):1566-72.
-
Prince JH, Diesem CD, Eglitis I, Ruskell GL. Anatomy and histology of the eye and orbit in domestic animals: Charles C. Thomas 1960.
-
Sinha RD, Calhoun ML. A gross, histologic, and histochemical study of the lacrimal apparatus of sheep and goats. Am J Vet Res 1966;27(121):1633-40.
-
Sundwall J. The structure of the Harderian glands of the ox. Anat Rec 1907;1:72–3.
-
Draper CE, Adeghate EA, Singh J, Pallot DJ. Evidence to Suggest Morphological and Physiological Alterations of Lacrimal Gland Acini with Ageing. Experimental Eye Research 1999;68(3):265-76.
-
Bromberg BB, Welch MH, Beuerman RW, Chew SJ, Thompson HW, Ramage D, et al. Histochemical distribution of carbonic anhydrase in rat and rabbit lacrimal gland. Investigative Ophthalmology & Visual Science 1993;34(2):339-48.
-
Draper CE, Adeghate E, Lawrence PA, Pallot DJ, Garner A, Singh J. Age-related changes in morphology and secretory responses of male rat lacrimal gland. Journal of the Autonomic Nervous System 1998;69(2-3):173-83.
-
Cornell-Bell AH, Sullivan DA, Allansmith MR. Gender-related differences in the morphology of the lacrimal gland. Investigative Ophthalmology & Visual Science 1985;26(8):1170-5.
-
Sullivan DA, Hann LE, Yee L, Allansmith MR. Age-and gender-related influence on the lacrimal gland and tears. Acta Ophthalmol (Copenh) 1990;68(2):188-94.
-
van Haeringen NJ. Aging and the lacrimal system. British Journal of Ophthalmology 1997;81(10):824.
-
McClellan KA, Robertson FG, Kindblom J, Wennbo H, Tornell J, Bouchard B, et al. Investigation of the Role of Prolactin in the Development and Function of the Lacrimal and Harderian Glands Using Genetically Modified Mice. Investigative Ophthalmology & Visual Science 2001;42(1):23-30.
Fig 1: Dorsal unipartite buffalo
lacrimal gland
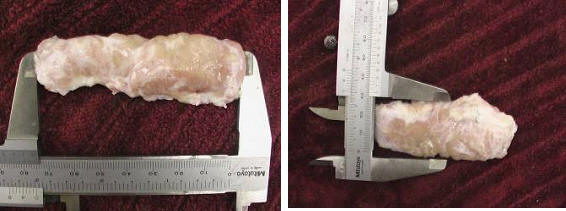
Fig 2 & 3:
Histological sections of the dorsal lacrimal gland of Iranian river buffalo.
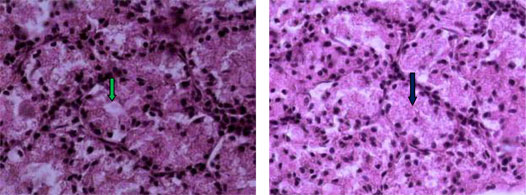
Note single sheets of
connective tissue separating the acinar and tubular units from each other and the tall pyramidal or columnar acini cells with small lumens. The buffalo acinar cells had basophilic, granular, vacuolated cytoplasm and appeared to be arranged in a loosely irregular fashion (black arrow).
The tubules have pseudostratified cells surrounding an empty lumen (green arrow).
Fig 4: Inter-lobular ducts with pseudostratified lining epithelium, veins and arterioles
|
 |





