|

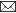
2010, Vol. 5 No. 1, Article 59
Oral Immunization of Mice with Rabies Vaccine
Using Different Baits
G. Salome1 and P. H. Ramanjini Gowda2*
1PG Scholar
2Professor and Head
Department of Biotechnology,
Vaccine Lab,
University of Agricultural Sciences,
GKVK, Bangalore
*Corresponding Author;
e-mail address: [email protected]
ABSTRACT
Rabies vaccine was used for oral immunization in mice with the locally available baits like sand, pieces of coconut shell, pieces of chickpea and wood pieces along with the additives like gram flour and jaggery solution. The efficient chewing of the baits by the mice and rupture of the buccal mucosa produced significantly higher levels of antibody titers.
KEY
WORDS
Rabies vaccine, Antibody titers, Bait, Oral immunization, Oral mucosal membrane.
INTRODUCTION
Rabies is a deadly communicable disease that is transmitted to a man from the rabid animals. It is estimated that 20,565 people die every year in India because of rabies (8). And most of the animal bites in India (91.5%) are by dogs, of which about 60% are strays and 40% are pets (6). Vaccination is the only way to control this disease but it is difficult to achieve hundred percent saturation vaccination of stray dog population through parenteral route only. So in addition to parenteral immunization, oral vaccination may be helpful in mass immunization program. Oral vaccines have proved to be effective in inducing immunity against rabies in wildlife (7).
Oral vaccination requires efficient baits that are hard enough to damage oral mucosal membrane on chewing so that the vaccine enters the blood stream through the ruptures.
MATERIALS AND METHODS
Vaccine: BHK – 21 cell-cultured inactivated animal rabies vaccine was used for testing in animals.
Animals:-6-8 weeks old Swiss Albino mice were used for the immunization studies. The mice were starved a day before oral immunization to increase its appetite.
Baits: Four different baits used for oral immunization studies included sand, pieces of coconut shell, pieces of chick pea and pieces of wood.. The coconut shell was used after removing the outer fiber and inner endosperm portion by grating. The shell of the coconut was broken into pieces of 2mm size. Chickpea (100 g) was grounded to 2mm size. Wood pieces were broken into small pieces of 2mm size and also 1mm sand was used.
Additives: Gram flour and jaggery was used as additive . Jaggery was crushed and dissolved in water to make jaggery solution.
Treatments:
The study was conducted on Six groups of(6-8weeks old) Swiss Albino mice with two in each group Two inoculation routes were used; intramuscular (IM) and oral feeding with different baits.
Group 1 : Prior to immunization the mice were fed with 40µl of 100 mg/ml of Sodium-bi-carbonate 15min to neutralize the acidity produced due to starvation Mice were fed orally with pieces of coconut shell (0.5g) + gram flour (1.5g) + 0.5 ml of animal rabies vaccine + Jaggery.
Group 2 : Mice were fed orally with sand (0.5g) + gram flour (1.5g) + 0.5 ml of animal rabies vaccine + jaggery.
Group 3 : Mice were fed with pieces of wood (0.5g) + gram flour (1.5g) + 0.5 ml of animal rabies vaccine + jaggery.
Group 4 : Mice were fed with pieces of chickpea (0.5g) + gram flour (1.5g) + 0.5 ml of animal rabies vaccine + jaggery.
Group 5 : Mice were fed with pieces of coconut shell (0.5g) + gram flour (1.5g) + 0.5 ml of animal rabies vaccine + jaggery.
During oral feeding, the feeding habit of mice was observed like initial contact with bait and chewing. The observation was done for 1-4 h.
The sixth group of mice was treated intramuscularly (IM) with 0.5ml of the animal rabies vaccine.
The inoculation was done on days 0, 7, 21 & 28.
Mice were bled by the retro-orbital route, after light ether anesthesia on day 40. Blood from each pair was mixed to obtain a sufficient quantity of serum. Serum was separated and heated at 56ºC for 30 min to destroy the heat-labile complement component and reduce contamination.
Estimation of antibody titres:
According to WHO recommended procedures Standard Mouse Neutralization Test (MNT) was performed for estimating neutralizing antibody titres in serum sample7. Different dilutions of each group serum were mixed with and LD50 of CVS of rabies virus and incubated in water bath @ 37ºC for 1h. The serum-virus mix was cooled and then inoculated intra-cerebrally into 6-8 week old mice, which were kept under observation for 14d. Depending on the percentage of survival and mortality of each dilution, the antibody titre was calculated and expressed in International units (IU) ml-1, compared to standard Rabies Immunoglobin (RIG).
Molecular analysis of Animal Rabies Vaccine:
SDS-PAGE:
Different concentrations (10-50µL) of animal rabies vaccine was resuspended in 1:1 (w/v) SDS-PAGE sample buffer [0.1M Tris-HCl, pH 6.9; 0.002% (v/v) Bromophenol blue; 2% (v/v) glycerol; 1% (w/v) SDS; 10% (v/v) 2-mercaptoethanol]. These protein samples were heated for 5-10 min at 100ºC and subjected to electrophoresis in 12% (w/v) SDS- Polyacrylamide gels. Gels were stained with Coomassie Brilliant Blue and destained, the protein bands were photographed.
RESULTS
Immunization of mice using animal rabies vaccine:
The titers of neutralizing (anti-glycoprotein) antibodies are summarized in Table 1. The mice which were treated intramuscularly with animal rabies vaccine gave higher antibody titers compared to oral treatments. Among the oral treatments, the mice fed with wooden pieces gave the lower titer compared to the other treatments.
Rabies Glycoprotein expression in animal rabies vaccine:
The animal rabies vaccine run on SDS-PAGE gel showed the presence of expected 66 kDa band at all concentration (10-50µl) (Figure. 1).
DISCUSSION
The immunization studies on mice with animal rabies vaccine have shown significant levels of antibody titers in all groups of mice. Group 1 mice which were fed with Sodium bi carbonate 15 min. prior to immunization to neutralize the acidity in stomach produced the same amount of neutralizing antibody as that of other treatment groups. Mice that were treated intramuscularly produced significantly higher levels of antibodies in comparison with the orally fed mice.
According to WHO, the antibody titer equal to or greater than 0.5 IU is sufficient to provide protection against infection by the rabies virus (9). The immunization study on mice has shown that the production of rabies neutralizing antibody was higher when treated intramuscularly (3.0 IU/ml). and significantly lower when immunized orally, that too was however more than the minimum antibody titer required to be produced (i.e., 1.5 IU/ml for protection against the disease. The mice, which were fed with wooden pieces, produced less neutralizing antibodies compared to other treatments of oral feeding but had sufficient neutralizing antibody titer(i.e., 0.5 IU/ml) and considered to give protection against rabies virus.
The results showed that, bait types used were efficient in providing oral immunization. The baits which are not chewed by an animal are less likely to be successful for oral vaccination, because vaccine contact with oral mucosal membrane would be limited and the vaccine may be inactivated due to stomach acidity (2). Similar study was done by Jayakumar et al. in dogs, in which they have shown that use of chicken heads as bait along with the vaccine made the dogs to chew the chicken head for a longer time and this lead to efficient distribution of vaccine in the oral mucous membrane4. Moreover, the hard baits disrupted the buccal cavity and the vaccine might have entered the blood stream providing efficient immunity.
The molecular weight of the rabies glycoprotein is found to be 66 kDa (3). The presence of 66 kDa bands in animal rabies vaccine shows that animal rabies vaccine sample is rich in rabies glycoprotein as thick band was seen even at 10 µL concentration of the sample. This is due to efficient glycosylation in animal cell culture system. The efficient glycosylation in animal and mammalian cell is due to the presence of terminal galactose and sialic acid residues (5).
ACKNOWLEDGEMENT
We thank Dr. Byre Gowda, Senior Scientist, Deputy Director of IAH&VB for kindly providing us the Animal Rabies Vaccine for our study.
REFERENCES
-
Atanasiu P. Quantitative assay and potency test of anti-rabies serum and immunoglobulin. Laboratory Techniques in Rabies, 3rd edn (WHO, Geneva, Switzerland) 1973; 314-316.
-
Baer G M, Broderson J R, Yager P A. Determination of the site of oral rabies vaccination. Am. J. Epidemiol 1975; 101:160-164.
-
Dietzschold B. Oligosaccharides of the glucoprotein rabies virus. Journal of Virology 1977; 23: 286-293.
-
Jayakumar R, Kumanan K, Nachimuthu K, Padmanaban V D. A bait delivery system suitable for oral rabies vaccine for stray dogs in India. Indian Vet. J 1998; 75: 207-210.
-
Ma J K C, Pascal M W D, Paul C. The production of recombinant pharmaceutical proteins in plants. Nature reviews 2003; 4: 794-805.
-
Menezes R. Rabies in India. CMAJ (Canadian Medical Association or its licensors) 2008; 178: 564-566.
-
Rupprecht C E, Wiktor T J, Johnston D H, Hamir A N, Dietzshold B, Wujnner W H, Glickman LT, Koprowski H. Oral immunization and protection of raccoons (Procyhon lotor) with a Vaccinia-rabies glycoprotein recombinant virus vaccine. Proceedings of the National Academy of Sciences of the USA 1986; 83: 7949-7950.
-
Sudarshan M K, Madhusudana S N, Mahendra B J, Rao N S N, Narayana A D H, Rahman A S, Meslin F X, Lobo D, Ravikumar K, Gangaboraiah. Assessing the burden of human rabies in India: results of a national multi-center epidemiological survey. Int. J. of Infectious Dis 2007;11: 29-35.
-
WHO. Expert Committee on rabies. Geneva (WHO Technical Report Series, 824) 1992.
-
WHO. Guidelines for research on oral rabies vaccines and field application of oral vaccination of dogs against rabies. Geneva 2007.
Figure 1
SDS-PAGE gel showing the presence of Rabies Glycoprotein in animal vaccine after staining with Coomassie Brilliant Blue. Lane M, marker; Lane 1, 10µL of animal vaccine; Lane 2, 20µL of animal vaccine; Lane 3, 30µL of animal vaccine; Lane 4, 40µL of animal vaccine; Lane 5, 50µL of animal
vaccine
Table 1:
Rabies virus neutralizing antibody titers (IU/mL) in mice immunized with animal rabies vaccine
|
Group
|
Mice Treatments
|
Neutralizing antibody titer
(IU/mL)
|
|
Group I
|
NaHCO3
+ Coconut shell+ animal vaccine
|
1.5
|
|
Group II
|
Animal
vaccine + Sand
|
1.5
|
|
Group III
|
Animal
vaccine + Wood pieces
|
0.5
|
|
Group IV
|
Animal
vaccine + Chickpea
|
1.5
|
|
Group V
|
Animal
vaccine + Coconut shell
|
1.5
|
|
Group VI
|
Treated
intramuscularly
with animal rabies vaccine alone
|
3.0
|
|
 |



