|
|
||||||||||
|
|
 |
|
|||||||||
|
|
|
 |
|
||||||||
|
|
|
||||||||||
|
|
|
||||||||||
|
|
|
||||||||||
|
|
|
||||||||||
|
|
|
||||||||||
|
2011, Vol. 6 No. 2, Article 87
Role of Kisspeptin in Puberty and Reproduction
Pratheesh M.D*, Radhika Syam1,
C. Harish2, R. Anoopraj2 and Justin Davis1
*Veterinary Physiology and Climatology Division
*Corresponding Author;
e-mail address: [email protected]
ABSTRACT
Kisspeptin (a product of the KiSS1 gene) and its receptor (GPR54) have emerged as key players in the regulation of reproduction in animals. Studies reported that kisspeptins stimulate the secretion of gonadotropins from the pituitary by stimulating the release of GnRH from the forebrain after the activation of GPR54, which is expressed by GnRH neurons. Kisspeptin is expressed abundantly in the arcuate nucleus (Arc) and the anteroventral periventricular nucleus (AVPV) of the forebrain. Kisspeptin neurons express the estrogen receptor and the androgen receptor, and these cells are direct targets for the action of gonadal steroids in both male and female animals, suggesting that kisspeptin signaling could mediate the neuroendocrine events that trigger the onset of puberty. Kisspeptin signaling in the brain has been implicated in generating the preovulatory GnRH/LH surge, triggering and guiding the tempo of sexual maturation at puberty, controlling seasonal reproduction, and restraining reproductive activity during lactation. Kisspeptin signaling may also serve diverse functions outside of the classical realm of reproductive neuroendocrinology, including the regulation of metastasis in certain cancers, vascular dynamics, placental physiology etc.
KEY
WORDS
Kisspeptin, GPR54, KiSS 1gene, puberty, AVPV nucleus,
arcuate nucleus.
INTRODUCTION
The kisspeptins (KP) were originally identified as a product of a metastasis suppressor gene, KiSS-1, in malignant melanomas by Lee et al (1996). Kisspeptins are endogenous ligands for a G-protein-coupled receptor named GPR54 (KiSS-1R). Proteolytic processing of the full-length 145 amino acid kisspeptin protein results in shorter fragments of the carboxyl -terminus region of the molecule with 54 (KP-54), 14 (KP14), 13 (KP-13) or 10 (KP-10) amino acids (Kotani et al. 2001). Initially, the largest cleavage product, KP-54 was identified for its ability to suppress the metastatic potential of malignant melanoma cells (Lee & Welch 1997) and it was therefore termed ‘metastin’.
TISSUE DISTRIBUTION AND LOCALIZATION.
Distribution of kisspeptin has been best described in rodents, where expression of both receptor (GPR54) and ligand (KiSS1) are the highest in placenta, with additional wide distribution throughout the central nervous system (the highest levels in hypothalamus and pituitary but also in cerebellum, cortex and brainstem) (Kotani et al. 2001). There are also some reports of variable expression in adipose tissue, pancreas, liver, small intestine, peripheral blood lymphocytes, testes, lymph nodes, and in human aorta, coronary artery and umbilical vein. (Muir et al. 2001, Hauge-Evans et al. 2006, Mead et al. 2007). In situ hybridization assays in mice have revealed that kisspeptin neurons (i.e. those that express Kiss1 mRNA) are most numerous in the arcuate nucleus (Arc), but significant numbers are also seen in periventricular nucleus (PeN) and the anteroventral periventricular nucleus (AVPV) (Smith et al.,2005). Although both sexes express Kiss1 mRNA in all five areas, expression in the AVPV is sexually dimorphic, where the expression of Kiss1 mRNA is much greater in females, the region directly implicated in the generation of pre-ovulatory GnRH/LH surge in female. This finding is not surprising considering that the AVPV in females is larger by volume and contains more neurons than the AVPV of males (Bleiber et al.,1982).
KISSPEPETIN REALLY TRIGGERS PUBERTY?
Does puberty begins with a ‘kiss’?. In mammals, activation of GnRH neurons is the key vent gating the onset of puberty; however, the mechanisms that trigger GnRH secretion at puberty remain one of the enigmas of modern science . Intriguingly, sexual maturation is impaired in animals with targeted deletions or spontaneous mutations in the GPR54 gene (Funes et al. 2003) suggesting that kisspeptin -GPR54 signaling is essential for pubertal maturation. Moreover, exogenous kisspeptin administered to prepubertal rodents and monkeys initiates various aspects of precocious puberty such as LH secretion or vaginal opening (Matsui et al.2004; Shahab et al.2005). Similarly, neural KiSS-1 gene expression increases in both male and female rats, mice and monkeys from pre- to post-puberty (Han et al. 2005); however, the specific KiSS-1 population(s) (ARC versus AVPV) that changes with puberty is equivocal and might reflect species-specific mechanisms that differ between primates and rodents (Plant et al.2005).Regardless, changes in the activity of KiSS-1 neurons appear to represent a seminal and perhaps common event in the timing of puberty in many species. Collectively, the findings indicate that hypothalamic-derived kisspeptin–GPR54–GnRH signaling is intimately involved in the mechanism(s) that initiate puberty, but precisely how this signaling is triggered remains a mystery.
THEORETICAL MODEL FOR THE FUNCTION OF KISSPEPTIN.
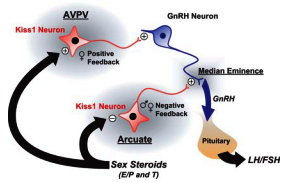
The enhancement of endogenous KiSS-1 tone in the hypothalamus takes place during pubertal attainment and such activation of GPR54 is apparently sufficient to trigger the neuroendocrine events leading to the onset of puberty. Kisspeptin stimulates the neuroendocrine reproductive axis, and sex steroids differentially regulate the expression of KiSS-1 mRNA in different nuclei within the forebrain. Kisspeptin released by neurons in the AVPV and Arc stimulates GnRH release, which induces the release of LH and FSH. The gonads respond to gonadotropins by secreting sex steroids, which then feed back to regulate the activity of kisspeptin neurons, inhibiting KiSS-1 expression in the Arc and inducing its expression in the AVPV (see Figure-1). The inductive effect of sex steroids on KiSS-1 expression in the AVPV may contribute to the preovulatory LH surge in females (and possibly T-mediated sex behavior in the male).
ROLE OF KISSPEPTIN IN VARIOUS PHYSIOLOGICAL PROCESSES IN ANIMALS.
1. Estrous cycle:
CONCLUSIONS
The pairing of the Kisspeptin with the GPR54 receptor has received growing attention since the description of the receptor as a molecular switch for puberty. Mechanisms underlying the role of GPR54 in puberty are the focus of the majority of emerging reports into this receptor system. A significant body of evidence across several species now suggests that KiSS-1 activation is a critical point in the commencement of puberty, although further investigation is required to fully characterize the interaction between Kisspeptin and the GnRH cascade.
REFERENCES
Bilban M, Ghaffari-Tabrizi N, Hintermann E, Bauer S, Molzer S, Zoratti C, Malli R, Sharabi A, Hiden U, Graier W. Kisspeptin-10, a KiSS- 1/metastin-derived decapeptide, is a physiological invasion inhibitor of primary human trophoblasts. Journal of Cell Science 2004; 117:1319–1328.
Bleier R, Byne W, Siggelkow I Cytoarchitectonic sexual dimorphisms ofthe medial preoptic and anterior hypothalamic areas in guinea pig, rat, hamster, and mouse. J Comp Neurol.1982; 212:118–130.
Castellano JM, Navarro VM, Ferna´ndez-Ferna´ndez R, Nogueiras R, Tovar S, Roa J, Vazquez MJ, Vigo E, Casanueva FF, Aguilar E, Pinilla L, Dieguez C, Tena-Sempere M Changes in hypothalamic KiSS-1 system and restoration of pubertal activation of the reproductive axis by kisspeptin in undernutrition. Endocrinology 2005; 146:3917–3925
Downs JL, Wise PM The role of the brain in female reproductive aging. Mol Cell Endocrinol. 2009 ;299:32–38.
Funes S, Hendrick JA, Vassileva G, Markowitz L, Abbondanzo S, Golovko A, Yang S, Monsma FJ and Gustafson EL. The KiSS-1 receptor GPR54 is essential for the development of the murine reproductive system. Biochem Biophys Res Commun. 2003 ;312: 1357–1363.
Gottsch ML, Cunningham MJ, Smith JT, Popa SM, Acohido BV, Crowley WF . A role for kisspeptins in the regulation of gonadotropin secretion in the mouse. Endocrinology 2004;145:4073–4077.
Han SK et al. Activation of gonadotropin-releasing hormone neurons by kisspeptin as a neuroendocrine switch for the onset of puberty. J. Neurosci. 2005; 25: 11349–11356
Hauge-Evans AC, Richardson CC, Milne HM, Christie MR, Persaud SJ & Jones PM A role for kisspeptin in islet function. Diabetologia ;2006: 49 2131–2135.
Horikoshi Y, Matsumoto H, Takatsu Y, Ohtaki T, Kitada C, Usuki S & Fujino M Dramatic elevation of plasma metastin concentrations in human pregnancy: metastin as a novel placenta-derived hormone in humans. Journal of Clinical Endocrinology and Metabolism. 2003;88 :914–919.
Janneau JL, Maldonado-Estrada J, Tachdjian G, Miran I, Motte N,Saulnier P, Sabourin JC, Cote JF, Simon B, Frydman R . Transcriptional expression of genes involved in cell invasion and migration by normal and tumoral trophoblast cells. Journal of Clinical Endocrinology and Metabolism 2002;87: 5336– 5339..
Kotani M, Detheux M, Vandenbogaerde A, Communi D, Vanderwinden JM, Le PE, Brezillon S, Tyldesley R, Suarez-Huerta N, Vandeput F. The metastasis suppressor gene KiSS-1 encodes kisspeptins, the natural ligands of the orphan G protein-coupled receptor GPR54. Journal of Biological Chemistry.2001;276: 34631–34636.
Lee JH & Welch DR Suppression of metastasis in human breast carcinoma MDA-MB-435 cells after transfection with the metastasis suppressor gene, KiSS-1. Cancer Research 1997;57: 2384–2387.
Lee JH, Miele ME, Hicks DJ, Phillips KK, Trent JM, Weissman BE & Welch DR. KiSS-1, a novel human malignant melanoma metastasis suppressor gene. Journal of the National Cancer Institute .1996; 88 :1731–1737.
Matsui H. et al. Peripheral administration of metastin induces marked gonadotropin release and ovulation in the rat. Biochem. Biophys. Res. Commun. 2004;320: 383–388
Mead EJ, Maguire JJ, Kuc RE & Davenport AP Kisspeptins are novel potent vasoconstrictors in humans, with a discrete localization of their receptor, G protein-coupled receptor 54, to atherosclerosis-prone vessels. Endocrinology.2007 ;148: 140–147.
Muir AI, Chamberlain L, Elshourbagy NA, Michalovich D, Moore DJ, Calamari A, Szekeres PG, Sarau HM, Chambers JK, Murdock P AXOR12, a novel human G protein-coupled receptor, activated by the peptide KiSS-1. Journal of Biological Chemistry 2001 ;276: 28969–28975.
Navarro VM, Castellano JM, Fernandez-Fernandez R, Barreiro ML, Roa J, Sanchez-Criado JE, Aguilar E, Dieguez C, Pinilla L and Tena-Sempere M Developmental and hormonally regulated messenger ribonucleic acid expression of KiSS-1 and its putative receptor GPR54 in rat hypothalamus and potent LH releasing activity of KiSS-1 peptide. Endocrinology 2004;145:4565–4574.
Plant TM. and Witchel SM. Puberty in non-human primates and humans. In Knobil and Neill’s Physiology of Reproduction (3rd edn) (Neill, J.D., ed.), Elsevier .2006: 2177–2230.
Revel FG, Saboureau M, Masson-Pe´vet M, Pe´vet P, Mikkelsen JD, Simonneaux V. Kisspeptin mediates the photoperiodic control of reproduction in hamsters. Curr Biol. 2006;16:1730–1735
Roa J, Aguilar E, Dieguez C, Pinilla L & Tena-Sempere M New frontiers in kisspeptin/GPR54 physiology as fundamental gatekeepers of reproductive function. Frontiers in Neuroendocrinology. 2008 ;29: 48–69.
Roa J, Vigo E, García-Galiano D, Castellano JM, Navarro VM, Pineda R, Die´guez C, Aguilar E, Pinilla L, Tena-Sempere M Desensitization of gonadotropin responses to kisspeptin in the female rat: analyses of LH and FSH secretion at different developmental and metabolic states. Am J PhysiolEndocrinol Metab. 2008a ;294:1088–1096 .
Shahab M. et al. Increased hypothalamic GPR54 signaling: a potential mechanism for initiation of puberty in primates. Proc. Natl Acad. Sci. USA. 2005; 102: 2129–2134
Smith JT, Coolen LM, Kriegsfeld LJ, Sari IP, Jaafarzadehshirazi MR, Maltby M, Bateman K, Goodman RL, Tilbrook AJ,UbukaT, Bentley GE, Clarke IJ,LehmanMN Variation in kisspeptin and RFamide-related peptide (RFRP) expression and terminal connections to gonadotropin-releasing hormone neurons in the brain: a novel medium for seasonal breeding in the sheep. Endocrinology 2008;149:5770–5782
Smith JT, Acohido BV, Clifton DK, Steiner RA. KiSS-1 neurones are direct targets for leptin in the ob/ob mouse. J Neuroendocrinol. 2006 ;18:298–303.
Smith JT, Dungan HM, Stoll EA, Gottsch ML, Braun RE, Eacker SM, Clifton DK, Steiner RA Differential regulation of KiSS-1 mRNA expression by sex steroids in the brain of the male mouse. Endocrinology 2005;146:2976–2984
Smith JT, Cunningham MJ, Rissman EF, Clifton DK, Steiner RA Regulation of Kiss1 gene expression in the brain of the female mouse. Endocrinology. 2005a; 146:3686–3692 .
Yamada S, Uenoyama Y, Kinoshita M, Iwata K, Takase K,Matsui H, Adachi S, Inoue K, Maeda KI, Tsukamura H Inhibition of metastin (kisspeptin-54)-GPR54 signaling in the arcuate nucleus-median eminence region during lactation in rats. Endocrinology 2007;148:2226–2232.
FIGURES
Fig 1 : A schematic representation of our current understanding of KiSS-1 signaling in the forebrain of the mouse. Kisspeptin stimulates GnRH secretion by a direct effect on GnRH neurons, most of which express the kisspeptin receptor, GPR54. Neurons that express KiSS-1 mRNA reside in the AVPV and the ARC (arcuate). KiSS-1 neurons in the ARC appear to be involved in the negative feedback regulation of GnRH/LH by sex steroids. The expression of KiSS-1 mRNA in the arcuate is inhibited by estradiol (E), progesterone (P), and testosterone (T). These same hormones induce KiSS-1 mRNA expression in the AVPV, where KiSS-1 neurons are thought to be involved in the positive feedback regulation of GnRH/LH.
Source: [Gottsch et al. 2004]
|
|
||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
|
|||||||||||
|
Copyright © Vet Scan 2005- All Right Reserved with
VetScan |
Home | e-Learning |Resources | Alumni | Forum | Picture blog | Disclaimer |
|
|||||||||
|
powered by eMedia Services |
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|