|

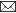
2011, Vol. 6 No. 2, Article 91
Anti-Diarrhoeal, Antimicrobial and Cytotoxic Effect
of Ethanol Extracted Guava (Psidium guajava) Leaves
Mohammad Mahmudul Hassan*1, A B M Shahinuzzaman1, Shahneaz Ali Khan1,
M B Uddin2 and Mohammad Mahabub-Uz-Zaman3
1Department of Physiology, Biochemistry and Pharmacology,
Chittagong Veterinary and Animal Sciences University, Khulshi, Chittagong-4202, Bangladesh
2Department of Medicine and Surgery, Sylhet Agricultural University, Sylhet-3100, Bangladesh
3Scientific Officer, Drug and Toxins Research Division, BCSIR Laboratory, Chittagong-4220, Bangladesh
*Corresponding Author;
e-mail address: [email protected]
ABSTRACT
Guava leaf (Psidium guajava) is commonly used to treat bacterial infection, gastroenteritis and diarrhoea. Present study was aimed at
determining the anti-diarrhoeal, antimicrobial and cytotoxic effect of the ethanol extracted guava plant leaves. The ethanol extracted guava (P. guajava) plant leaves showed significant (P<0.05) anti-diarrhoeal activity on castor oil induced diarrhoea in model albino rat comparing with loperamide (P<0.01). The leave extract tested against ten pathogenic bacteria and exhibits prominent antibacterial activity against five gram positive (Stayphylococcus aureus, Bacillus polymyxa, Bacillus cereus, Bacillus megnatum and Bacillus subtilis) bacteria and one gram negative (10.5mm). bacteria with highest zone of inhibition(13.25mm) for Proteus sp followed by Stayphylococcus aureus (10.5mm). LC50 of ethanol extract of guava leaves against brine shrimp nauplii was 828.7μg/ml and the wide range of LC50 value denoted the safety effect of the extract.
KEY WORDS
Anti-diarrhoeal, antimicrobial, cytotoxicity, ethanol extracted guava leaf, albino rat.
INTRODUCTION
Guava (Psidium guajava) is useful medicinal plant its leaves have been used in folk medicine for many years to treat diarrhoea, stomach ache and hepatic problems. In vitro antibacterial activity of Guava leave extract against diarrhoea causing bacteria like Staphylococcus, Shigella, Salmonella, Bacillus,
E. coli, Pseudomonas has been reported by Gran and Demello, (1999).. The bark, leaves, fruit and root have also been evaluated pharmacologically for the treatment of gastrointestinal diseases (Tona, 1999.). This plant possesses antimicrobial, anticough, haemostasis (Jaiarj et al., 1999; 2000), anti mutagenic (Grover and Bala, 1993), hypoglycaemic (Raman et al., 1995), antidiarrhoeal and narcotic (Lozoya et al., 1990), antioxidant (Qian and Nihorimbere, 2004) properties. The leaf extract has also been used to treat diarrhoea, abdominal pain, convulsions, epilepsy, cholera, insomnia and hypertension (Lutterodt, 1989; Lutterodt and Maleque, 1988 and Nacoulma, 1996. Present study was taken up to evaluate antimicrobial, anti-diarrhoeal and cytotoxic effect of ethanol extract guava leaves.
MATERIALS AND METHODS
Preparation of plant extracts
Sample collection
Fresh Guava (Psidium guajava) leaves collected from the Bangladesh Agricultural Research Institute, Chittagong (BARI) were chopped into small pieces air dried for 21 days. Dried leaves were ground into fine powder and stored in air tight containers in a cool, dark and dry place until use.
Extraction
3000 ml of ethanol was poured gradually into a clean glass jar containing 500g of dried leave powder. The lid was closed properly to resist the entrance of air into the jar. The jar was kept for 15 days with regular stirring by means of a clean glass rod at 2-3 days of interval.
Filtration
The leaf extract was filtered through sterilized cotton filter rinsed with ethanol. The filtrate was collected in a beaker and filtrated again through whartman filter paper. The filtrate thus obtained was evaporated to dry by vacuum evaporative Rotatory machine (Model 2).
Re-Extraction
Dried material was subjected to re-extraction in 500 ml ethanol. The filtration procedure was repeated and 51.98g leaves extract was obtained and kept and stored at 4ºC.
Animal and diet
Albino rats weighing on average 150-200g were selected from Animal house of Chittagong Veterinary and Animal Sciences University, The animals were acclimatized to
standard lab conditions (temperature 24±1ºC , relative humidity 55±5% and 12 hrs photoperiod) for one week before the commencement of the experiment in suspended wire meshed galvanized cages (4-6 rats/cage). During the period of study rats were supplied with a semi- purified basal diet adlib water and maintained according to the NIH guidelines for care and use of Laboratory Animals (Saha et al, 2001)
Anti-diarrhoeal Assay
Test protocol (Castor oil induced diarrhoea)
15 albino rats were allotted to three (control, positive control and test group) groups. 2ml water, 2ml plant extract and 2ml of anti-diarrhoeal (loperamide) per rat were administered orally to control, positive control and test group respectively. After half-an hour 2ml of castor oil per rat was orally administered to each group to induced diarrhoea (Yegnanaryan and Shostri, 1982). Faecal material was checked for consistency every hour. was defined as the Presence of fluid material in the stool that stained the absorbent paper placed beneath the cage indicated induction of
diarrhoea. The number of respondents and stool passed during 6 hrs period was recorded for each rat to determine the effect of plant extract against diarrhoea.
Disc Diffusion Method for Preliminary Assay of Antimicrobial Effect (Bauer et al, 1966).
Pure cultures of 10 bacterial organisms ( Staphylococcus aureus BTCC:43 ,
Shigella sommei ICDDR‟B , Klebsilla sp ICDDR‟B ,
Bacillus polymyxa BTCC:16 , Bacillus cereus BTCC:19 ,
Bacillus megatenusm BTCC:18 , Bacillus subtilis BTCC:17 ,
Salmonella typhi ICDDR‟B , Proteus sp ICDDR‟B ,
Shigella flexinary ICDDR‟B.) were collected from the microbiology section of BCSIR laboratory. One loop of pure culture of each organisms was inoculated into individual test tube containing nutrient broth and incubate at 37ºC for 24hrs to develop sub culture of each of the ten organisms .These cultures were streaked individually on ten nutrient agar plates and incubated at 37ºC for 24hrs to promote bacterial multiplication throughout the
petridish.
A solution of 100μg/ml concentration of guava leave extract was prepared by dissolving the extract in alcoholic solvent. Sterilized and dried filter paper discs (4mm in diameter) were impregnated with leave extract solution using micropipette.. Three discs, one each of leave extract, tetracycline(30μg/ml). and solvent soaked were placed on each agar plate seeded with specific bacterial culture . All plates were incubated at 37ºC for 24hrs to allow the maximum growth of the organisms. The antimicrobial effect of guava leave extract was determined by comparing the diameter of “Zone of Inhibition‟. of leave extract disc with that of tetracycline disc.
Cytotoxicity Assay
The cytotoxicity assay was performed on brine shrimp nauplii using Brine-Shrimp Lethality Assay (Meyer et at., 1982). Brine shrimp nauplii were obtained by hatching brine shrimp eggs (Carolina Biology Supply Company, Burlington, NC, USA) in artificial sea water (22.5g sodium chloride dissolved in 500ml water) for 48hrs. Dissolution of guava leave extract was performed in artificial sea water. 5ml solution of different concentrations (62.5, 125, 250, 500, 1000, 2000 μg/ml) of the leave extract were taken in seperate beakers containing brine shrimp nauplii and observed for mortality for 24hrs. Artificial sea water medium containing DMSO was taken as control. The data were transformed to probit analysis for the determination of LC50 values of the extract.
Statistical Analysis
Data were expressed as Mean ± S.E. (Standard Error). Unpaired “t” tests were applied for significant tests. SPSS for WINDOWS@TM Statistical Package for social science for Windows was applied for analysis of data. Probability (P) value of 0.05 or less (P≤0.05) was considered as significant. In case of cytotoxicity, the LC50 values were calculated by “Probit Analysis”.using “Bio-State Software-2007”
RESULTS AND DISCUSSION
Anti-diarrhoeal assay
The anti-diarrhoeal assay showed that the leave extract protected diarrhoea up to the level of 55.6%. Whereas the protection level of diarrhoea by loperamide was 75%. . The mean latent period of defecation in leave extract treated group was 3.2± 1.31.hours much shorter than control group (7.2± 0.49). Leave extract had a significant (P<0.05) anti diarrhoeal effect. However, the effect was less than that of loperamide (P<0.01). These findings were in accordance with the observation of Gosh and Sen, (2006) and John et al., (2008) .In Rural areas of Bangladesh Guava leaves are usually chewed to check diarrhoea and also relieve discomfort and pain associated it. Lutterodt, (1989), reported similar observations by using the leaves, bark of the stem and root of the
P. guajava.
Antimicrobial assay
Out of 10 bacterial organisms 6 were found sensitive to the leave extract and 9 to tetracycline. The antibacterial activity of Guava leave extract against various Gram ‘+’ve and Gram ‘-‘ve organisms was earlier reported by Francise and Heine, (2006); Goncalves et al., (2005). Higher sensitivity of
Staphylococcus aureus and resistance of Salmonella and Shigella to Guava leave extract reported during present trial was in close agreement with the findings of Lutterodt and Ismail, (1999).
Cytotoxicity assay
The LC50 value of guava leave extract found 828.7(μg/ml) in this study that indicates the plant leaves extract of guava (Psidium guajava) have wide range of LC50 value.
Cytotoxic activity of Guava leave extract
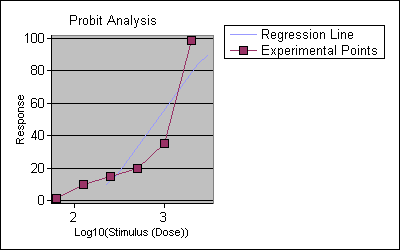
Percentage mortality of brine shrimp at 62.5, 125, 250, 500, 1000, and 2000 μg /ml concentrations of leave extract was observed to be 0%, 5%, 10%, 35%, 65% and 100% respectively .From the Percentage mortality of brine shrimp, the probits were calculated for each concentration. Probits were then plotted against corresponding leave extract log concentration and from the plot LC50 (log concentration 50) value was 2.9 μg /ml. The LC50 value of leave extract was found to be 828.7 μg /ml with 95% confidence limit .The lower and upper limits were 316.1, 1024.2 μg /ml respectively. The wider LC50
values of leave extract indicate that its toxicity level in minimum.
The value of LC50 for Amorphophallus campanulatus plant determined by using Brine-Shrimp Lethality Assay was found to be 7.66 μg /ml and was interpreted as a toxic plant Alam et al., (2007).
Conclusion
Ethanol extract of guava leaf has efficient level of anti-diarrhoeal and antimicrobial effect. As its LC50 value is wider which indicates its safety margin and indicates that its toxicity level in minimum.
REFERENCES
-
Alam K, Moizur R, Shariful I. Antibacterial, Antifungal and Cytotoxic Activities of Tuberous Roots of Amorphophallus campanulatus. Turk J Biol 2007; 31:167-172.
-
Bauer AW, Kirby E, Sherris EM, Turk M. Antibiotic by standarized single disk method. Am. J. Clin. Path1966; 45: 493-496.
-
Francis P, Heine E. Antimicrobial activity of Psidium guajava on selected bacteria, Masters thesis, Universiti Putra, Malaysia, 2006, pp134.
-
Gnan SO, Demello MT. Inhibition of Staphylococcus aureus by aqueous Psidium guajava leaves extracts. J Ethnopharmacol1999; 68:103-108.
-
Gonçalves JLS, Lopes RC, Oliveira DB, Costa SS, Miranda MMFS, Romanos MTV, Wigg MD. In vitroanti-rotavirus activity of some medicinal plants used in Brazil against diarrhea. J Enthopharmacol2005; 99:403-407.
-
Gosh TK, Sen T. Antimicrobial activity of guava (Psidium guajava Linn.) leaf extract on diarrhea causing enteric bacteria. J Enthpharmaco, 2006;7:431-433.
-
Grover IS, Bala S. Studies on antimutagenic effects of guava (Psidium guajava) in Salmonella typhimurium. Genetic Toxicol, 1993; 300:1-3.
-
Jaiarj P, Khoohaswan P, Wongkrajang Y, Peungvicha P, Suriyawong P, Saraya MLS, Ruangsomboon O. Anticough and antimicrobial activities of Psidium guajava Linn. leaf extract. J Ethnopharmacology, 1999; 67:203-212.
-
Jaiarj P, Wongkrajang Y, Thongpraditchote S, Peungvicha P, Bunyapraphatsara N, Opartkiattikul N. Guava leaf extract and topical haemostasis. Phytother Res, 2000; 14:388-391.
-
John OA, Emmanuelo A, Witness DH. Antidiarreal activity of Psidium guajava Linn. (Myrtaceae leaf aqous in rodents) J smooth muscle research, 2008; 37:195-207.
-
Lozoya X, Bercerril G, Martinez M. Intraluminal perfusion model of in vitro guinea pig ileum as a model of study of the antidiarrheic properties of the guava (Psidium guajava). Arch Invest Med (Mex), 1990; 21:155–156.
-
Lozoya X, Meckes M, Abou-Zaid M, Tortoriello J, Nozzolillo C, Amason J.T. Quercetin glycosides in Psidium guajava L. leaves and determination of spasmolytic principle. Arch Med Res, 1994; 25:11-15.
-
Lutterodt GD, Maleque A. Effects on mice locomotor activity of a narcotic-like principle from Psidium guajava leaves. J Ethnopharmacology, 1988; 24:219-231.
-
Lutterodt GD. Inhibition of gastrointestinal release of acetylcholine by quercetin as a possible mode of action of Psidium guajava leaf extracts in the treatment of acute diarrhoeal diseases. J Ethnopharmacology, 1989; 25:235-247.
-
Lutterodt GD, Ismail. Antimicrobial effect of Psidium guajava extract as one mechanism of it’s antidiarrhea action . J Med Sci, 1999; 6 (2):17-20.
-
Meyer BN, Ferrigni NR, Putnam JE, Jacobson LB, Nichols DE, McLaughlin JL. Brine shrimp: a convenient general bioassay for sactive plan constituents. Planta Medica, 1982; 45: 31-34.
-
Nacoulma O. Plants medicinales et pratiques medicales traditionelles au Burkina Fasdo, University de Ouagadougon, 1996 ; 2:285.
-
Qian H, Nihorimbere V. Antioxidant power of phytochemicals from Psidium guajava leaf. J Zhejiang Univ Sci, 2004; 5(6):676–683.
Raman R, Flores S, Alarcon F. Plant use for medicine purpose. J Enthopharmacol, 1995; 56:81-87.
-
Saha BK, Sarkar A, Basak R, Chatterjee M. $ 25-dihydrooxycholecalciferol (Vitamin D3) suppresses the effect of streptozotocin- induced diabetes during chemical rat liver carcinogenesis, Cell Biology international (London), 2001; 25(3): 227-237.
-
Seshadri TR, Vasishta K. Polyphenols of the leaves of Psidium guajava: quercetin, guaijiaverin, leucocyanidin and amritoside. Phytochem, 1965; 4:989–992.
-
Tona L. Biological screening of traditional preparations from some medicinal plants used as antidiarrhoeal in Kinshasa, Congo. Phytomedicine, 1999; 6:59–66.
-
Yegnanarayan R, Shostri DS. Comparison of antidiarroheal activity of sons drugs in experimental diarrhea, Indian J. pharmacol, 1982; 14(4): 293-299.
TABLES
Table 1:
Details of organisms isolated from vaginal discharge of infertile bitches and their antibiogram pattern
|
Group
|
Treatment
|
Dose (Concentration)
|
Rat with diarrhoea
|
%
of protection
|
Mean defecation within 6hrs
|
%
inhibition of defecation
|
|
Control
|
Distilled water
|
2ml/rat
|
5/5
|
0%
|
7.2± 0.49
|
-
|
|
Positive control
|
Loperamide
|
2ml/rat
(2mg/ml)
|
3/5
|
40%
|
1.8± 1.11
|
75%
|
|
Treated
|
Ethanol
Extracted P.
guajava
leaves
|
2ml/rat (200mg/ml)
|
4/5
|
20%
|
3.2± 1.31
|
55.6%
|
Table 2:
Comparison of effect of Guava leave extract and loperamide on mean defecation of albino rats using Student’s t-test
|
Mean defecation (6 hrs study period)
|
|
Control
|
Loperamide
(2
mg/ml)
|
P.
guajava
leaves extract
(200
mg/ml)
|
|
7.2 ±
0.49
|
1.8 ±
1.11 **
|
3.2 ±
1.21*
|
|
Student’s t test
|
t
tabulated
|
3.35
|
2.57
|
|
degrees of freedom
|
8.0
|
8.0
|
|
P
value
|
<0.01
|
<0.05
|
Table 3:
Comparison of zones of inhibition (mm diameter)
|
Name
of the organism
|
Tetracycline
(30µg/disc)
|
P. & guajava leaf
extract (100µg/disc
|
|
Staphylococcus aureus BTCC:43
|
16.5
|
10.5
|
|
Shigella sonnei ICDDR’B
|
7.5
|
-
|
|
Shigella flexneri ICDDR’B
|
7
|
-
|
|
Bacillus polymyxa BTCC:16
|
15.5
|
7.5
|
|
Bacillus cereus BTCC:19
|
10.5
|
8.5
|
|
Bacillus subtilis BTCC:17
|
10
|
9
|
|
Bacillus megaterium BTCC:18
|
12
|
8
|
|
Proteus sp
ICDDR’B
|
-
|
13.25
|
|
Salmonella typhi ICDDR’B
|
14.5
|
-
|
|
Klebsilla spp
ICDDR’B
|
28
|
-
|
Table 4:
Brine shrimp lethality bioassay of ethanol extract of Guava leaf (P. guajava)
|
Dose (µg/ml)
|
Log dose
|
Total
|
Alive
|
Death
|
%
(Lethality)
|
Actual
%
|
Probit
|
|
62.5
|
1.80
|
20
|
20
|
0
|
0
|
0.0125
|
2.116
|
|
125
|
2.10
|
20
|
18
|
2
|
10
|
0.1
|
4.099
|
|
250
|
2.40
|
20
|
17
|
3
|
15
|
0.15
|
3.975
|
|
500
|
2.70
|
20
|
16
|
4
|
20
|
0.2
|
4.192
|
|
1000
|
3.00
|
20
|
13
|
7
|
35
|
0.35
|
4.616
|
|
2000
|
3.30
|
20
|
0
|
20
|
100
|
0.9875
|
6.569
|
Table 5:
Calculation of LC50 value, regression equation and confidence limit by probit analysis
|
Log10 LC50
|
LC50(μg /ml)
|
95% confidence (μg /ml)
|
Regression equation
|
Chi-square
|
|
2.9
|
828.7
|
316.1-1024.2
|
Y=-1.5754+2.2889*X
|
6.37
|
FIGURES
Figure 1. Percentage of various isolates obtained on culturing vaginal discharge from infertile bitches
Figure 2. Regression line and Experimental point
|
 |




